Taro leaf blight
Pacific Pests, Pathogens and Weeds - Online edition
Pacific Pests, Pathogens & Weeds
Taro leaf blight (014)
Phytophthora colocasiae
Asia, Africa, North (Hawaii), South (Argentina, Brazil) America, the Caribbean, Oceania. It is recorded from American Samoa, Guam, Federated States of Micronesia, Northern Mariana Islands, Palau, Papua New Guinea, Samoa, and Solomon Islands. The disease spread to West Africa in 2010.
The fungus attacks taro and Alocasia macrorrhizos; these are the only hosts known from the Pacific islands.
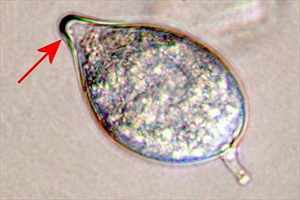
Phytophthora colocasiae is a water mould, an oomycete, not a fungus. It is related to algae. Spots occur on the leaves, and often these have yellow margins (Photo 1). After a few days, a white ring can be seen at the margin of the spots (Photo 2); this is the area where large numbers of spores (Photo 3) are produced during the night. However, the spores dry out quickly in the sun and by mid-morning they have shrivelled and died. They only stay alive if it is cloudy or raining. The next night another ring of spores will form.
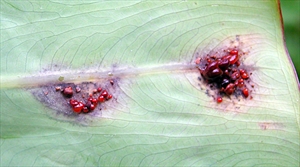
Underneath the spots, red-brown merging droplets occur (Photos 4). The droplets dry as dark pellets (Photo 5).
Infection occurs anywhere on the leaf surface, but often at the edges where rain and dew collect. Spots occur on the leaf stalks (petioles) of very susceptible varieties.
The spots merge and secondary spots develop (Photo 6), and rapidly a blight develops as most of the leaf blade become infected and begins to die.
Spread of the water mould occurs in a number of ways: (i) wind and rain to other leaves, nearby plants and distant plantations; (ii) on suckers planted with infected leaves attached; (iii) as infections on leaf stalks of planting material (called 'tops') in two ways, (a) as spots on the petioles of susceptible varieties, and (b) as infections at the cut ends of leaf stalks when they are trimmed for planting.
When spores (called 'sporangia') land on the surface of a leaf they can germinate in two ways:
- They germinate directly, like a seed.
- The spores produce smaller spore inside them, up to 20; these are called ('zoospores'). The sporangia burst and release the zoospores which swim about for a short while, then settle down, germinate and infect.
In either cases, infections cause the brown spots.
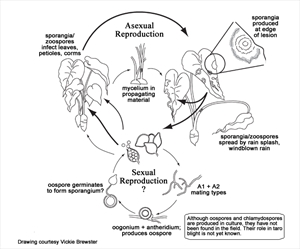
The water mould exists as two strains: A1 and A2. If the two forms mate an 'oospore' is produced which has thick walls and can survive times when the crop is not growing. However, only the A2 strain occurs in the Pacific island countries (Diagram).
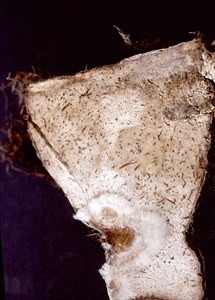
Corms become infected after harvest, when the suckers are removed (Photo 7).
Taro leaf blight reduces the life of the leaves: instead of lasting 40 days they last 20. The result is that plants have few leaves and, consequently, produce small corms. Yield is reduced by about 40%.
Taro leaf blight also causes a firm, brown corm rot after harvest. Corms become completed decayed by the water mould in 7-10 days, often with fungi present in the rots helping the decay process. (see Fact Sheet no. 179).
Look for spots that grow rapidly, join together and cause a blight. Look for plants with leaves that die early - infected plants have 3-4 leaves instead of the 6 -7 on those that are healthy. Look for the white rings of spores at the margin of the spots, seen most clearly in the early morning. Look for dark, irregular-shaped pellets on the underside of the spots.
QUARANTINE
Note, that there are some countries in the Pacific still vulnerable to this disease, so where it is not yet present, biosecurity authorities should be alert to potential pathways for its introduction. If countries want to introduce germplasm for testing, they should follow the FAO/IBPGR Technical Guidelines for the Safe Movement of Edible Aroid Germplasm (http://www.bioversityinternational.org/e-library/publications/detail/edible-aroid/). This stipulates that introductions should be as virus-tested plants growing in tissue culture.
CULTURAL CONTROL
Where rainfall is high, taro leaf blight is difficult to control. Cultural methods can be tried, but they are often not effective.
Before planting:
- Make new gardens as far away as possible from old ones. It is best if the gardens are surrounded by forest.
- Do not plant gardens downwind from older gardens with the disease.
- If possible, make gardens in the highlands, above 500 m The disease is less at cooler temperatures.
- Do not plant suckers with leaves attached; the leaves may be infected.
During growth:
- Inspect young taro twice a week; cut off infected leaves as soon as they are seen. Do this regularly; however, if rainfall is high, it can only delay the disease.
After harvest:
-
Harvest when the leaves are dry; otherwise, spores may infect the ends of the leaf stalks when they are cut as 'tops' for the next crop.
- To prevent storage rots, wash the soil off the corms, trim the leaves and put them in plastic bags. Washing corms in 1 per cent household bleach before placing them in plastic bags improves storage.
RESISTANT VARIETIES
Breeding programs in Papua New Guinea and Samoa have produced plants resistant to taro leaf blight. These are available from the CePaCT lab of the Secretariat of the Pacific Community. In Solomon Islands, a hybrid, LA16, has been bred for resistance to taro leaf blight.
CHEMICAL CONTROL
-
Use copper fungicides (especially copper oxychloride). However, sprays are needed every 10-14 days, and are only recommended where taro are grown for sale. Application using a motorised knapsack sprayer is more efficient and effective than using a hydraulic knapsack sprayer.
- Use metalaxyl (a systemic product) in combination with copper; it gives even better control, but costs are high.
- Use phosphorous acid (or potassium phosphonate) alternating with mancozeb (used successfully in Samoa).
____________________
When using a pesticide, always wear protective clothing and follow the instructions on the product label, such as dosage, timing of application, and pre-harvest interval. Recommendations will vary with the crop and system of cultivation. Expert advice on the most appropriate pesticide to use should always be sought from local agricultural authorities.
AUTHORS Helen Tsatsia & Grahame Jackson
Information from Jackson G (2020) Taro leaf blight - My 50-year part in its downfall. (https://www.pestnet.org/the-taro-leaf-blight-story/); and Carmichael A, et al. (2008) TaroPest: an illustrated guide to pests and diseases of taro in the South Pacific. ACIAR Monograph No. 132, 76 pp. (https://lrd.spc.int/about-lrd/lrd-project-partners/taropest); and Gollifer DE, Brown JF (1974) Phytophthora leaf blight of Colocasia esculenta in the British Solomon Islands. Papua New Guinea Agricultural Journal 25: 6-11; and Singh D, et al. (2012) Taro Leaf Blight - A Threat to Food Security. Agriculture 2: 182-203; and Third taro symposium. Edited by Guarino L, et al. (2003) Report of a meeting (technical). Secretary of the Pacific Community. 242 pp; and from Tyson JL, Fullerton RA (2007) Mating type of Phytophthora colocasiae from the Pacific region, India and South-east Asia. Australasian Plant Disease Notes 2: 111-112. Photos 1&6 Brooks FE (2005) Taro leaf blight. The Plant Health Instructor. DOI:10.1094/PHI-I-2005-0531-01. Diagram Vickie Brewster.
Produced with support from the Australian Centre for International Agricultural Research under project PC/2010/090: Strengthening integrated crop management research in the Pacific Islands in support of sustainable intensification of high-value crop production, implemented by the University of Queensland and the Secretariat of the Pacific Community.