Coconut scale, coconut false scale
Pacific Pests, Pathogens, Weeds & Pesticides - Online edition
Pacific Pests, Pathogens, Weeds & Pesticides
Coconut (false) scale (348)
Aspidiotus rigidus; previously Aspidiotus destructor rigidus.
Indonesia and the Philippines. It has not been recorded in Oceania.
Coconuts, and other palms, including ornamentals. It also occurs on banana and mangosteen.
The adults and nymphs occur in large masses on lower leaf surfaces, blocking air exchange, and stopping photosynthesis, and sucking the sap (Photo 1). Infestations cause leaves to yellow, wilt and dry out (Photos 2&3). Nuts are affected, too: the 'meat' is less and the milk is sour. Heavy infestations cause seedlings and young palms to die.
Eggs (about 12) are laid in a crescent shape under one half of the cover or 'armour' of the female (Photo 4). These hatch producing nymphs called 'crawlers'. Soon, they settle and form the armour, using the cast skins. More moults occur until the scale is mature, and the armour is oval to circular, 1.5-3 mm across, yellow to brown, tough and transparent. The female body, under the armour, is pale or greyish-greenish. Mature males are yellow, have wings, eyes and legs, but no mouthparts. It is not certain whether they are needed to mate with the females or eggs develop without fertilisation.
The scale prefers high humidity and is most often found on the lower side of leaves. Similarly, outbreaks of the scale are favoured in dense stands of coconuts, especially if growing in semi-wild shady conditions.
Spread occurs when crawlers disperse over short distances, or longer when carried by wind currents, vehicles, animals, birds, or on clothing. All stages are spread in the horticultural trade of plants or plant parts. The life cycle is about 45-55 days.
Since the beginning of the outbreak in the Philippines in 2009, spread of the scale has been rapid and severe. Trees have died and without a practical control measure farmers have lost incomes, resorting to cutting down dying trees and selling the wood. By 2013, the scale had spread to nearly 800,000 trees, and spread is continuing. Even moderate attack can cause copra losses of 25-50%.
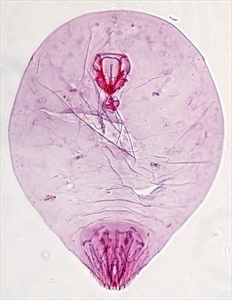
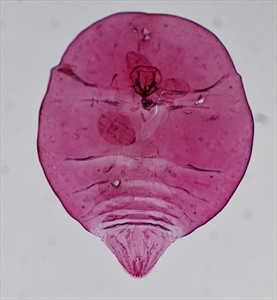
This coconut (false) scale is very similar to the coconut scale, Aspidiotus destructor (see Fact Sheet no. 104). The scale cover is tougher and more rigid than that of the Aspidoptus destructor, and there are body differences but, to see these, slide-mounted specimens need to be examined by an expert (Photos 5&6). There is also a difference in the length of the life cycle which is 1.5 times longer in Aspidiotus rigidus, and egg skins are laid in a crescent under half of the scale armour. The egg skins of Aspidiotus destructor are all around the scale (Photo 7).
QUARANTINE
The narrow distribution of this scale requires biosecurity organisations to take precautions against its further spread. The FAO/IBPGR Technical Guidelines for the Safe Movement of Coconut Germplasm should be followed when imports of coconuts are made (see, (http://www.bioversityinternational.org/e-library/publications/detail/coconut/). It is particularly important that they are followed as the scale has a limited host range and there is no effective method of biological control.
NATURAL ENEMIES
The predatory beetles, Chilocorus nigrita, Chilocorus melas and Telsimia nitida have been found associated with coconut scales but, seemingly, not controlling outbreaks. Wasp parasitoids have been recorded in Indonesia (North Sulawesi), e.g., Spanioterus crucifer, Aphytis chrysomphali, and Comperiella unifasciata, but these appear to have little impact. It is possible, from studies in Bali, that Encarsia citrina attacks Aspidiotus rigidus. By contrast, the scale has been present in Java for some time without causing outbreaks. but it is not known if natural enemies are keeping it under control. It is possible that several predators and parasitoids are required to manage this pest. Searches in Java are needed.
However, in 2017 studies in the Philippines showed that a Comperiella species parasitises 65-90% of the Aspidiotus rigidus, and palms recovered where the parastoid was present. In late 2017, this wasp was being multiplies and distributed in the Zamboanga region of Mindanao.
IPM - THE PHILIPPINES' APPROACH
The situation in the outbreak areas is not sufficiently clear to provide management options. Gillian Watson has reported that outbreaks in most, but not all, of the Philippines, end naturally after 2 years because of reduced production of offspring, and high mortality for reasons unknown.
Present recommendations of the Philippines Coconut Authority follow an IPM strategy which includes leaf pruning, insecticides - trunk injections and sprays -biocontrol agents, and fertilizer to hasten recovery. It remains to be seen it this cocktail of actions provides a practical solution. In the long-term, however, biological control is likely to be the only viable option.
The Fertilizer and Pesticide Authority has granted emergency use approval to Crop Guard (Chemrez Technologies), a product made from vegetable oil.
Recommendation provide for the coconut scale Aspidiotus destructor (see Fact Sheet no. 104) are as follows:
CULTURAL CONTROL
In the early stages of infestations, remove fronds colonised by the coconut scale, and check seedlings for scale infestations before planting.
RESISTANT VARIETIES
None known.
CHEMICAL CONTROL
Insecticides may be useful on young palms, but not those that are mature because the crowns are too high to reach. There is also the likelihood that insecticides will destroy predators and parasitoids and delay bringing about natural and sustained control of the coconut scale.
- If necessary to use an insecticide, use white oil (made from vegetable oils), soap solution, or horticultural oil (made from petroleum) (see Fact Sheet no. 56).
- White oil:
- 3 tablespoons (1/3 cup) cooking oil in 4 litres water.
- ½ teaspoon detergent soap.
- Shake well and use
- Soap:
- Use soap (pure soap, not detergent).
- 5 tablespoons of soap in 4 litres water, OR
- 2 tablespoons of dish washing liquid in 4 litres water.
- White oil:
- Commercial horticultural oil can also be used. White oil, soap and horticultural oil sprays work by blocking the breathing holes of insects causing suffocation and death. Spray the undersides of leaves; the oils must contact the insects. A second application of soap or oils may be necessary after 3-4 weeks.
- The addition of malathion is useful against scales insects, but it is likely to kill natural enemies. It is best avoided, if possible.
- Synthetic pyrethroid insecticides are likely to be effective against the crawlers - crawlers are the active nymphs that spread infestations, but they are difficult to see as they are so small. And synthetic pyrethroids are likely to kill natural enemies. They are best avoided, if possible.
____________________
When using a pesticide, always wear protective clothing and follow the instructions on the product label, such as dosage, timing of application, and pre-harvest interval. Recommendations will vary with the crop and system of cultivation. Expert advice on the most appropriate pesticides to use should always be sought from local agricultural authorities.
AUTHOR Grahame Jackson
Information from Watson GA, et al. (2016) Aspidiotus rigidus Reyne (Hemiptera: Diaspididae): a devasting pest of coconuts in the Philippines. Agriculture and Forest Entomology 17:1-8. (https://www.researchgate.net/publication/264159039_Aspidiotus_rigidus_Reyne_Hemiptera_Diaspididae_A_devastating_pest_of_coconut_in_the_Philippines#:~:text=The%20coconut%20scale%20insect%2C%20Aspidiotus,Philippines%20between%202010%20and%202015); and CABI (2020) Aspidiotus rigidus. Crop Protection Compendium; and from Molet T (2015) CPHST Pest Datasheet for Aspidiotus rigidus. USDA-APHIS-PPQ-CPHST (http://download.ceris.purdue.edu/file/2821). Photos 1-4 Merle Shepard/Gerald Carner, Clemson University, USA. Photos 5-7 Plant Pest Diagnostics Branch, California Department of Food & Agriculture, Sacramento, USA.
Produced with support from the Australian Centre for International Agricultural Research under project PC/2010/090: Strengthening integrated crop management research in the Pacific Islands in support of sustainable intensification of high-value crop production, implemented by the University of Queensland and the Secretariat of the Pacific Community.