Fall armyworm. Named after its flight into North America where it lays eggs, and the larvae develop in the Fall or Autumn.
Pacific Pests, Pathogens, Weeds & Pesticides - Online edition
Pacific Pests, Pathogens, Weeds & Pesticides
Fall armyworm (401)
Spodoptera frugiperda
Asia, Africa, North, South and Central America, the Caribbean, Oceania. As of April 2021, it has reached Australia and Papua New Guinea. It is native to North and South America.
More than 80 species. The armyworm has a preference for plants in the grass family, the Poaceae, i.e., maize, millets, rice, sorghum, sugarcane, but it also attacks banana, cotton, cowpeas, ginger, peanut, tobacco, some forage legumes, and vegetables, e.g., beans (including soybean), cabbage, capsicum, cauliflowers, cucumber, eggplant, potato, sweetpotato, tomato, and some weeds and ornamentals. There are two types: one prefers maize and sorghum; the other, rice and forage (pasture) grasses.
Damage is done by the larvae (Photos 1&2). On young plants, the larvae eat the leaves except for the waxy layer resulting in a symptom known as 'windowing'; seedlings may also be cut at the base. On older plants, holes are made in the leaves, edges are eaten, and ears (cobs) of maize are bored (Photo 3). Damage to the cobs may be much more serious than damage to the leaves.
Eggs are laid in masses (120-200) on the underside of lower leaves, covered in scales from the female. Up to 10 batches may be laid by a single moth. Young larvae are green, with dark heads and dark lines alongside the body; at first they feed together skeletonising leaves and making 'windows'. As the larvae develop - there are six stages taking 14-21 days in total - they become brown with three whitish lines along the back, develop dark spots with spines, and an upside-down Y on the head. When mature, larvae are 4-5 cm long. They also become cannibalistic; on maize, only 1-2 larvae survive per whorl. Larger larvae are nocturnal unless migrating in swarms.
Pupation is underground, inside a loose cocoon made from silk and soil; more rarely it occurs between leaves on the host. Pupation lasts about 12 days. Adults have a wingspan of 32-40 mm, with a brown or grey forewing and white hindwing (Photo 4).
The moth is a stronger flyer and travels fast; with wind assistance some travel up to 1000 km in a lifetime. Spread also occurs via the international trade in vegetables and ornamentals.
Spread across Africa was fast; since its first detection in 2016, it has been reported in 28 countries, and tens of millions of hectares of maize have been infested, plus large areas of sorghum and millet. Estimated losses from sub-Saharan Africa are put between US$2.5-6.2 billion a year. Similar rapid spread has occurred across India beginning in 2018, and in January 2019 the armyworm reached Sri Lanka, and several Southeast Asian countries.
Impact is considerable because early larvae require little food whereas later stages require a lot; this means that early infestations are often missed, and outbreaks are seen only when the armyworm matures. It may then be too late to prevent serious damage. However, though visual damage can often be considerable (e.g., voracious feeding of maize whorls), this regularly is reflected in relatively low to moderate economic impacts, i.e., 10-20% reduction in crop yields.
In Africa (and elsewhere), pesticide-based control is often ineffective as the moth was introduced from the Americas with resistance to carbamate, organophosphorus, organochlorine, and pyrethroid insecticides. Ample cost-effective and practical options exist for non-chemical control of fall armyworm, including biological control and cultural means.
Look for the inverted Y on the front of the larvae (the head capsule) and four dark spots in a square on the last but one segment. However, as there are several Spodoptera species, confirmation of identification ideally needs to be done by specialists. There are morphological keys and molecular methods available. Once identification has been done, use the app developed by FAO (FAMESWS) for Android mobile devices to standardise monitoring in the field or when using pheromone traps. Scouting information is added to a global database to track movement of the pest (http://www.fao.org/fall-armyworm/en/), and help improve management. The app also has information on IPM and biological control.
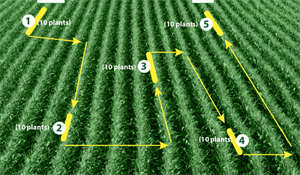
FAO Guidance Note 2 (http://www.fao.org/fall-armyworm/faw-management/pesticide-guidance/en/) shows how to sample a small field (2 ha or less). Walk a letter 'W' covering the entire field, selecting a group of 10 plants at the beginning and then at each turn so that there are five groups (see Diagram). Count the number of infested plants in each group. An infested plant is where there is fresh damage or fresh frass on the leaves or in the whorls of leaves (maize). Add the number of infested plants, and calculate a percentage (x2 total). FAO Guidance Note 3 (same URL) describes trapping using a pheromone.
BIOSECURITY
Increasing transportation and international trade are likely to facilitate the further spread of the fall armyworm outside Africa and Asia. Australia, Indonesia, Malaysia, the Philippines and Pacific island countries are at high risk from the spread of Spodoptera frugiperda. However, although a quarantine issue, it is also a biosecurity problem. The moth is high flying and so it is impossible to stop its spread.
Moths caught in the vortices of cyclones in the southern tropical cyclone belt from Africa to Southeast Asia could deliver the moth to Australia and Papua New Guinea.
NATURAL ENEMIES
Several wasps have been identified from larvae at different locations. Those of most interest are Cotesia marginventris, Chelonis insularis, Trichgramma pretiosum, and Telenemus remus. Telenomus remus, an egg paraitoid, gives control in the Caribbean and is now of interest in Africa. These and a fly, Winthemia trinitatis, are mentioned in the FAMEWS app. Predators include, birds, rodents, beetles, earwigs and other insects - ants, ladybird beetles, earwigs, predatory stink bugs, and others.
The avoidance of synthetic pesticides & diversification of corn fields can help conserve many of the above natural enemies and contribute to pest control. Additionally, there are fungi, bacteria and viruses that infect the armyworm and cause diseases. Death from pathogens is about 10-15%. Foliar sprays of a pest-specific nucleopolyhedrosis virus (NPV) can be a highly-effective option for the field-level management of fall armyworm. NPV inoculates can be collected in the field by farmers, and stored in the fridge for future application.
CULTURAL CONTROL
Before planting:
- Plant crops early to avoid increases in fall armyworm as the season progresses.
- Use high quality seed, and fast-growing seedlings.
- Plant all cereal crops at the same time to avoid overlapping crops and movement of the fall armyworm from older to younger crops.
- If not possible to plant all at one time, avoid planting new crops next to old, especially where larvae are present.
- Use push-pull intercropping.
- The 'pull' - plant a trap crop, e.g., napier or Brachiaria grasses between rows of maize - the moths lay their eggs on the grasses rather than maize, but the larvae do not survive.
- The 'push' - Desmodium is planted between rows of maize. This improves the soil and is said to repel Spodoptera frugiperda moths.
- Interplant with e.g., cassava which is not a favoured host of the fall armyworm, or use legume intercrops (e.g., peanut, cowpea) to diversify the cropping system and enhance overall health of the maize plants. The establishment of intercrops also helps diversify income sources, and moderates economic losses due to pest attack.
During growth:
- It is important to visit the crop frequently and regularly, at least once a week. See Detection & Inspection for methods of crop monitoring.
- If scouting indicates Spodoptera frugiperda is present and requires control, do the following:
- Crush egg masses by hand.
- Remove the larvae by hand if only a few are present, and the planted area is small (less than 2 ha); if the area is larger or if infestation of larvae is high, consider using a botanical or biopesticide. But consider carefully which ones to use (see below).
- Alternatively, for small plots, place sand or ash in the whorls of maize, or use diluted sugar-water to attract ants to infested maize plants. See the FAO FAMEWS app for details of these home remedies. (https://play.google.com/store/apps/details?id=org.fao.famews&hl=en_US).
- Plant flowering trees, shrubs and herbaceous ornamentals to provide nectar for parasitoids.
- Provide perches for birds.
- Provide adequate nutrients, by applying synthetic or organic fertilizers. Vigorously growing plants are better able to replace damaged leaves.
After harvest:
- Collect and bury as much of the crop as possible.
RESISTANCE VARIETIES
There are varieties of maize with resistance due to tougher leaves. GM varieties are available that produce Bt (Bacillus thuringiensis) toxins.
CHEMICAL CONTROL
Experience in Africa suggest that gains from using insecticides are rarely economically justified for resource-poor smallholders. For maize at least, it is difficult for insecticides to reach the armyworm when deep in the whorl and the entrance is covered with frass, plus the fact that yield loss is rarely more than 20%, and often nearer 10%. There is also the harm to natural enemies, biodiversity and human health to consider. However, if pesticides are needed, do the following:
- For small plots, use plant-derived pesticides (PDPs), such as neem, derris, pyrethrum and chilli (with the addition of soap).
- Note, Derris, brought many years ago to Solomon Islands from Papua New Guinea, is effective as a spray. It contains rotenone, an insecticide, so it should be used with caution. There may be varieties of Derris (fish poisons) in your country that can be tried (see Fact Sheet no. 56).
- For larger plots where PDPs are often inappropriate use commercial biopesticides that contain disease-causing organisms, such as:
- Bt - Bacillus thuringiensis subspecies kurstaki (strain SA-11). Bt is best against young larvae.
- Beauveria bassiana (strain R444).
- Spodoptera frugiperda multiple nucleopolyhedrovirus (SfNNPV).
- Note, organophosphate, organochlorine, carbamate and synthetic pyrethroid products are unlikely to be effective in the long run: (i) the fall armyworm already has resistance to many kinds, or will develop resistance quickly, and (ii) these insecticides will destroy natural enemies, and make the crop more susceptible to pest attack.
____________________
When using a pesticide, always wear protective clothing and follow the instructions on the product label, such as dosage, timing of application, and pre-harvest interval. Recommendations will vary with the crop and system of cultivation. Expert advice on the most appropriate herbicides to use should always be sought from local agricultural authorities.
AUTHOR Grahame Jackson
Information from John Wightman (2019) Fall armyworm update. Natural Resource Development Maleny Queensland. (https://api.pestnet.org/media_store/submissions/2019/4/8855f10f-95cc-4b1c-8d78-1ed14562de1a/Fall%20armyworm%20note.pdf); and CABI (2019). Spodoptera frugiperda (fall armyworm) Crop Protection Compendium. (https://www.cabi.org/cpc/restricted/?target=%2fcpc%2fdatasheet%2f29810); and Fall armyworm. Wikipedia. (https://en.wikipedia.org/wiki/Fall_armyworm); and FAO FAMEWS V3 app; and FAO Fall Armyworm Guidance Notes 1-5. (http://www.fao.org/fall-armyworm/faw-management/pesticide-guidance/en/); and from Kris Wyckhuys. Hanoi, Vietnam. Photo 1 Russ Ottens, University of Georgia, Bugwood.org. Photo 2 Frank Peairs, Colorado State University, Bugwood.org. Photo 3 John C. French Sr., Retired, Universities:Auburn, GA, Clemson and U of MO, Bugwood.org. Photo 4 Lyle Buss, University of Florida, Bugwood.org. Diagram from FAO FAW Guidance Note 2 Fall Armyworm Scouting. (http://www.fao.org/3/I8321EN/i8321en.pdf).
Produced with support from the Australian Centre for International Agricultural Research under project HORT/2016/18: Responding to emerging pest and disease threats to horticulture in the Pacific islands, implemented by the University of Queensland and the Pacific Community