Yam dry rot. It is also known as yam nematode, yam dry rot nematode.
Pacific Pests, Pathogens, Weeds & Pesticides - Online edition
Pacific Pests, Pathogens, Weeds & Pesticides
Yam tuber dry rot nematode (529)
Scutellonema bradys.
Asia (India, Pakistan), Africa, North, South and Central America, the Caribbean. It is NOT recorded from Oceania.
Dioscorea species. The main species are Dioscerea rotundata-cayenensis, Dioscorea alata, Dioscorea dumentorum, Dioscorea esculenta, and others. The following in other plant families are also main hosts: cowpea, melon, and sesame.
Dry rot of yam caused by Scutellonema bradys is a serious disease of all edible and wild species. Symptoms are seen only at harvest, but the dry rots, aided by secondary fungal infections, continue to increase in storage. The disease is similar to that caused by Pratylenchus coffeae, a common nematode of yam and other crops in Oceania (see Fact Sheet no. 8).
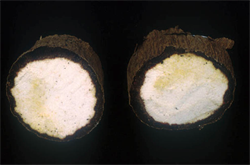
Feeding of the nematodes produces shallow areas of rot beneath the skin; initially they are cream to yellowish, turning light brown and finally dark brown, dry and powdery. Normally, the areas of rot are less than 2 cm deep beneath the surface, but often extend around the tuber (Photo 1). Externally, cracks appear in the skin, which becomes crinkly, and parts flake off revealing the dark rot beneath (Photos 2&3). Rots continue in storage and can lead to complete decay of the tuber as other rot-causing organisms take over. There are no above ground symptoms.
Sometimes external symptoms are not obvious on the tubers. Extensive rots can develop without cracking and flaking of the skin, and are only seen when the skin is scraped away.
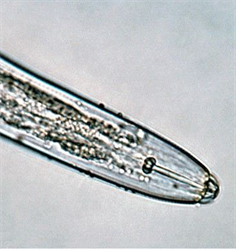
The nematode enters alongside roots and shoots as they emerge from tuber sets, and also cracks or damaged areas in the skin. They have a hollow spear which they use to break cells and suck up the contents (Photo 4). They have a hollow needle-like mouthpart, called the spear, which they inject into cells of the tuber and feed on their contents. Both males and females live in the tubers and soil around them; eggs are laid and the young develop into adults after several moults.
Spread of Scutellonema over short distance is in water within and above ground, and in soil on tools and shoes. However, the main method of spread over short and long distances is in sets used for planting. Survival of the nematode occurs on the roots of other crops and weeds, and in stored tubers.
The nematode causes four main problems on yams: (i) a reduction in the weight of diseased tubers, up to 30% less than healthy ones at harvest; this is due to moisture loss, and is worse in late harvested tubers in dry soil; (ii) lower quality tubers that reduces their market value; (iii) a loss of edible portions, which increases the longer the yams are stored; and (iv) a loss of planting material. Long-term losses are estimated to be 50%; when wet rots in storage follow dry rots, losses can be 80-100%.
Look for tubers at harvest with crinkly, scaly skin covering shallow brown dry rots. Surveys of tubers from markets in West Africa across the yam belt, where the disease is common, found highest populations of the nematode in Dioscora rotundata in the mid-altitude savannah, followed by the southern Guinea savannah. In Nigeria, these surveys found that approximately half the tubers at markets are infested with the nematode.
BIOSECURITY
The risk of introduction is high, especially associated with the trade in yam tubers. Small numbers of nematodes may be present in yams after harvest without noticeable signs of their presence. For this reason, and also because there is evidence of different strains of Scutellonema bradys the unrestricted movement of varieties of yam from one country to another should be prohibited, and transfer made only as sterile, pathogen-tested plants, following the FAO/IBPGR (1989) Technical Guidelines for the Safe Movement of Yam Germplasm.
CULTURAL CONTROL
Before planting:
-
Carefully check tubers for dry rot symptoms by scraping away small areas of skin; it is easier to check for dry rot if tuber cuttings or mini-sets are used for planting rather than whole tubers.
-
Wipe the knife used to cut the sets with a cloth soaked in bleach, especially if the knife has cut infested tubers.
-
Coat the sets in wood ash; even it will not significantly reduce nematode numbers, it may add beneficial potassium.
- Avoid planting yams in fields where previous crops were any of the following alternative hosts of the nematode: cowpea, sesame, green gram, pigeon pea, kenaf (Hibiscus cannabinus), okra, tomato and melon. Preference should be given to maize, groundnut, chilli, Indian spinach or sorghum.
During growth:
-
Ensure healthy growth. Add cow dung to the mound to reduce nematode populations, or add applications of NPK fertilizer, but nitrogen alone may increase populations.
-
Keep weeds to a minimum, especially those that are hosts of yam nematode, e.g., Eupatorium, Synedrella and Chromolaena.
After harvest:
-
Collect all undersized and rotten tubers after harvest and bury deeply or burn.
-
Ideally, plant yams after a fallow period; this is often difficult as pressure on land is intense and increasing; nor is it always obvious to farmers what crops to use as Scutellonema has a wide host range. However, restorative fallows using legume cover crops reduce nematode populations and the following are recommended: Aeschynomene histrix, Pueraria phaseoloides, Mucuna puriens and Centrosema species.
RESISTANT VARIETIES
No varieties of either Dioscora cayenensis-rotundata or Dioscorea alata with nematode resistance are yet available. The main methods of controlling dry rot are by cultural methods, the most important of which is the use of clean, nematode-free planting material. Nematode resistance is an object of the yam breeding program at the International Institute of Tropical Agriculture.
CHEMICAL CONTROL
This is not an appropriate method for the control of this disease as most nematicides are dangerous to use without training, they are expensive and mostly unavailable to smallholders. By contrast, hot water treatment is inexpensive and beneficial. Yams should be immersed in water at 51°C for 10 minutes. As a thermometer is needed to ensure the water is kept at the required temperature, this may not be a viable method for smallholders unless they are trained. If used, farmers should test it on a few sets first, as a method to produce nematode-free planting material for the next season’s crop. Treatment should be applied near the end of dormancy, but before shoots develop, not immediately after harvest.
AUTHOR Grahame Jackson
Information from CABI (2021) Scutellonema bradys (yam nematode). Crops protection Compendium. (https://www.cabi.org/cpc/datasheet/49315); and Bridge J, et al. (2005) Nematode parasites in tropical root and tuber crops (excluding potatoes). In: Luc M, Silora RA, Bridge J (eds) Plant parasitic nematodes in subtropical and tropical agriculture (second edition). CABI Publishing, Wallingford, UK, pp 221-258. (http://books.google.com.au/books?id=GAdsEt6dEtwC&pg=PA250&lpg=PA250&dq=scutellonema+bradys+yam&source=bl&ots=hM7P6PcHyY&sig=RoHp8WUtDqAEunoy9quvOSG_xfA&hl=en&sa=X&ei=e7bgU_nIMoPq8AX_jIKIBQ&ved=0CBsQ6AEwADgK#v=onepage&q=scutellonema%20bradys%20yam&f=false); Brunt AA, et al. (1989) FAO/IBPGR Technical Guidelines for the Safe Movement of Yam Germplasm. Food and Agriculture Organization of the United Nations, Rome/International Board for Plant Genetic Resources, Rome. (http://ecoport.org/Resources/Refs/IPGRI/yam.pdf); and from Yam nematode (Scutellonema bradys). Plantwise Technical Factsheet. (http://www.plantwise.org/knowledgebank/datasheet.aspx?dsid=49315). Photo 1 Dry rot of yam. IITA. (http://bit.ly/1FV6y4g). Photos 2&4 John Bridge Damage symptoms on yam tubers. CABI. (https://www.cabi.org/cpc/datasheet/49315). Photo 3 Yam tuber infected with dry rot. IITA. (http://bit.ly/1LKoMgW).
Produced with support from the Australian Centre for International Agricultural Research under project HORT/2016/185: Responding to emerging pest and disease threats to horticulture in the Pacific islands, implemented by the University of Queensland, in association with the Pacific Community.