Black rot
Pacific Pests, Pathogens, Weeds & Pesticides - Online edition
Pacific Pests, Pathogens, Weeds & Pesticides
Sweetpotato black rot (232)
Ceratocystis fimbriata; the asexual state is Chalara species.
Asia, Africa, North, South Central America, Europe, Oceania. Ceratocystis fimbriata is recorded from Australia, American Samoa, Fiji, New Zealand, Papua New Guinea, Samoa, and Solomon Islands. In Papua New Guinea, the fungus is assocated with stem and storage roots.
The fungus attacks a wide range of annual and perennial plants other than sweetpotato, for example: wattle, pigeon pea, Alocasia, cassava, citrus, coffee (canker), cocoa (wilt), carrot, mango (blight), pineapple (see Fact Sheet no. 190), rubber (mouldy rot), sugarcane (black rot, see Fact Sheet no. 218), taro (black rot), Xanthosoma, and forest trees. There are many strains of the fungus and they have different host ranges. Some infect only the host from which they were found, whereas others infect several other hosts. There are some strains from sweetpotato that only infect sweetpotato, and the same applies to strains from taro, rubber and coffee. Molecular studies have placed the strains into three groups: North America, Latin America and Asia.
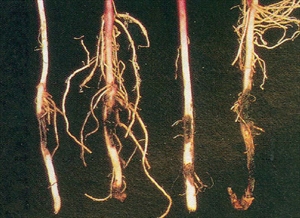
The fungus is a wound pathogen. Small, circular, slightly sunken, grey or dark brown, spots occur over the surface of the storage roots, darkening with age (Photos 1&2). Symptoms on the storage roots appear either before or after harvest. Spots also occur on the stems below ground causing them to wilt, turn yellow and die (Photo 3).
Survival of the fungus occurs as a special type of spore ('ascospore') that has thick walls so it can survive in soil. The cottony growth of the fungus also survives inside storage roots after harvest, the length of time extended if the roots are maintained in cold storage. It also survives in the vines and discarded storage roots left in the field.
Spread of the fungus occurs as spores brought to wounds, either natural or man-made, in the wind or by insects. The fungus produces a strong fruity smell when grown on artificial media; if this smell also occurs in nature, it may attract insects. Some beetles that feed on fungi and plant sap bring spores to wounds in the storage roots made by soil-living insects, by rats, and by the process of harvest. The spores are sticky and beetles become covered in them. It is also likely that the fungus is spread on bags and baskets used at harvesting, and on tools and farm machinery.
Spread long distances also occurs in storage roots (sweetpotato) and corms (taro) which are traded, and also used as planting material.
The disease on sweetpotato is important in many parts of the world, although losses are not well reported. In the US, damage is now less severe in the southeastern states than previously, mostly due to the use of resistant varieties and hygiene measures. However, it remains an important disease in China and Japan.
Look for the black, circular, slightly sunken rots on the storage roots; these are characteristics of the disease, although there may be black rots on the stems, and the stems may wilt.
QUARANTINE
The unrestricted movement of plant propagating material (cuttings, shoots and storage roots) has the potential of further spreading the fungus, and should be done with caution. It could also be moved internationally in logs and wood chips used for fuel, paper, horticulture, packing material and other uses. There are FAO/IBPGR Technical Guidelines for the Safe Movement of Sweet Potato Germplasm (http://www.bioversityinternational.org/e-library/publications/detail/sweet-potato/).
CULTURAL CONTROL
Before planting:
- If you are using a nursery to grow plants for cuttings, make sure that this is on 'new' land, and not on sites that have been used for sweetpotato in the past. And make sure that the cuttings or storage roots used to start the nursery are free from black rot symptoms.
- When taking cuttings, either from the nursery or the field, always use tip cuttings, about 30-35 cm long. Never take back cuttings with roots attached or underground parts of the stem.
During growth:
- Be careful to handle the storage roots carefully at harvest to avoid wounding the storage roots.
After harvest:
-
Remove the remains of the crop after harvest, and destroy.
-
Rotate crops, leaving a 3-4-year interval between crops of sweetpotato planted on the same land. Do not plant taro or Xanthosoma after sweetpotato as these crops are susceptible to infection by the black rot fungus. Instead, plant banana, brassicas, cucumber, ginger, maize, peanuts and other food legumes, e.g., shallots, solanaceous crops, or yams.
-
Do not wash and package storage rots from crops that show signs of the disease.
-
Do not wash healthy storage roots and then store them wet; allow them to dry first in a well-ventilated place.
RESISTANT VARIETIES
There is no information on differences in the tolerance of varieties in the Pacific to this disease.
CHEMICAL CONTROL
Fungicides have been suggested for the treatment of cuttings before planting in order to kill spores of the fungus. Should a fungicide be needed, use thiabendizole.
____________________
When using a pesticide, always wear protective clothing and follow the instructions on the product label, such as dosage, timing of application, and pre-harvest interval. Recommendations will vary with the crop and system of cultivation. Expert advice on the most appropriate pesticide to use should always be sought from local agricultural authorities.
AUTHOR Grahame Jackson
Information from Amante V, et al. (2003). A field guide to the sweetpotato problems in the Philippines. The University of Queensland (https://www.soilwealth.com.au/imagesDB/news/Sweet-Potato-Pest-and-Disease-Guide.pdf); and O'Sullivan J et al . Sweetpotato DiagNotes: A diagnostic key and information tool for sweetpotato problems. (http://keys.lucidcentral.org/keys/sweetpotato/key/Sweetpotato%20Diagnotes/Media/Html/FrontPage/FrontPage.htm); and from Nelson SC (undated) Black rot of sweet potato: disease cycle and management. CTAHR, Hawaii. (https://www.ctahr.hawaii.edu/nelsons/BlackRotofSweet%20Potato[2].pdf). Photo 1 Gerald Holmes, California Polytechnic State University at San Luis Obispo, Bugwood.org. Photo 2 Charles Averre, North Carolina State University. Bugwood.org. Photo 3 Compendium of SweetPotato Diseases (1988). American Phytopathological Society.
Produced with support from the Australian Centre for International Agricultural Research under project PC/2010/090: Strengthening integrated crop management research in the Pacific Islands in support of sustainable intensification of high-value crop production, implemented by the University of Queensland and the Secretariat of the Pacific Community.